Stanford Professor Emeritus Brock-Utne Pens New Article but Doesn’t Disclose More Than $48,000
For years, we’ve been examining how industry-funded pundits, masquerading as unbiased experts, are published and aired by influential media.
So when we received a tip last October about Physicians Against Drug Shortages and its media campaign against entities that purchase medical supplies in bulk known as Group Purchasing Organizations (GPO), we decided to take a look. That trail led us eventually to Stanford University Professor of Anesthesia (Emeritus) John G. Brock-Utne, MD, PhD.
Known internationally, Dr. Brock-Utne wrote an article for the November/December 2018 issue of The Bulletin, a publication of the Santa Clara County Medical Association and the Monterey County Medical Society, in which he made the case that “drug makers and other suppliers have been locked out of the hospital marketplace by group purchasing organizations.” The same argument has been made by certain drug and medical device companies.
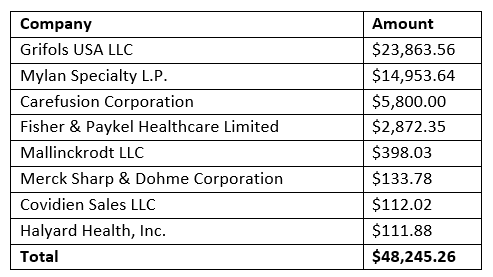
Dr. Brock-Utne is certainly entitled to his opinions. However, what he didn’t mention, nor did The Bulletin, is that Dr. Brock-Utne has received more than $48,000 from drug and medical device companies since 2013, according to the Centers for Medicare and Medicaid Services Open Payments database. Not mentioned, as well, is that Dr. Brock-Utne is a co-chair of Physicians Against Drug Shortages (PADS).
An Obligation to Disclose?
While not connected to PADS, the lack of financial disclosure has been highlighted with another physician. A recent joint investigation by the New York Times and ProPublica into the former chief medical officer of Memorial Sloan Kettering Cancer Center, Dr. José Baselga, reported that he had failed to accurately disclose payments from health care companies in dozens of research articles. Subsequently, Dr. Baselga resigned from Sloan Kettering, as one of the chief editors of the journal Cancer Discovery and from the boards of three drug and equipment makers. The NYT-ProPublica story reported:
“Baselga’s disclosure lapses have prompted a broader discussion over the influence of the drug and health care industries…Teaching hospitals across the country have reminded faculty members of their obligation to disclose, and some have begun re-examining which relationships are appropriate.”
Questions for Brock-Utne
We’ve sent an email to Dr. Brock-Utne asking the following questions:
- In light of the recent New York Times-ProPublica investigation into the influence of healthcare companies on medical journal research, should you have disclosed the more than $48,000 you received since 2013 from drug and medical device companies when you submitted your article to The Bulletin?
- Do you see the $48,000 you received from drug and medical device companies as a financial conflict of interest?
- Will you ask The Bulletin to update your article with more disclosure of your ties to drug and medical device companies?
- Have you coordinated your message in The Bulletin with Physicians Against Drug Shortages Executive Director Phillip Zweig or any other PADS co-chair?
- Are you aware of any financial contributions from drug or medical device companies to Physicians Against Drug Shortages?
We’ll update our readers once we hear back.
PADS Undisclosed Ties
In a similar manner, shouldn’t others associated with Physicians Against Drug Shortages disclose their conflicts?
PADS Executive Director Phillip Zweig, for example, had past ties to Retractable Technologies, Inc. (RTI), where he was employed as communications director and received stock options. RTI appears to have a long-history of opposing GPOs, which they blame for the drug shortage. Zweig has been asserting that same position since 2012, when PADS was established.
Co-Founder Zivot
PADS traces its origins to a conversation between Zweig and Dr. Joel Zivot, the Chief of Cardiothoracic Intensive Care at Emory University Medical Center, according to the PADS website.
In 2014, PADS Co-Founder and Co-Chair Joel Zivot received $2,000 from the Masimo Corporation, “a global medical technology company that develops and manufactures innovative noninvasive patient monitoring technologies, medical devices, and a wide array of sensors.” The payment was described as a speaking, training or education engagement. Zivot has, in the past, participated in the media campaign.
Other PADS co-chairs have received sums from drug makers and other suppliers, but do not appear to have contributed to the group’s ongoing media campaign. They include:
Co-Chair Atkins
Since 2015, PADS Co-Chair Joshua Atkins, M.D. of Philadelphia has received more than $66,000 from drug and medical device companies. The largest source of payments was from Beckton Dickinson —nearly $49,000 in Associated Research Funding and General Payments.

Co-Chair Bernard
Since 2013, PADS Co-Chair Dr. Rebekah Bernard from Fort Myers, Florida received more than $4,000, according to the Centers for Medicare and Medicaid Services Open Payments. The largest single source of payments was more than $500 from Janssen Pharmaceuticals, Inc.
Other PADS Co-Chairs
Also since 2013, PADS Co-Chair Dr. Sean Adams from Naperville, Illinois received more than $1,000 and Co-Chair Dr. Michelle Abe from Martinsburg, West Virginia received more than $800. All of these payments from drug and medical device companies were described as Food and Beverages, Travel and Lodging or Education.
Send Us a Tip
PADS claims to have no conflicts of interest, no vested financial interest in the issue of drug shortages and no outside funding. The financial ties of these PADS co-chairs call these claims into question. If you have information to share about other payments or financing, please send us a confidential or totally anonymous tip by clicking on the link at the top of our website or HERE. We protect all sources.
Scott Peterson is executive director of Checks and Balances Project, an investigative watchdog blog that seeks to hold government officials, lobbyists and corporate management accountable to the public. Funding for C&BP is provided by Renew American Prosperity and individual donors.
You May Want to Read:
Why Does Physicians Group Promote Kaleo – a Company Investigated by the U.S. Senate and 60 Minutes?
Phillip Zweig Disrupts Health Policy Forum on Root Causes of Drug Shortages
Physicians Against Drug Shortages Get a Pass from 16 Media Outlets in Our Survey







Recent Comments